|
Back to Blog
Ion bonding service5/3/2023 Pendant cationic and anionic groups are randomly distributed along polyelectrolyte chains by random polymerization of ionic monomers. Sacrificial ionic bonds in hydrogels can be formed between oppositely charges polyelectrolyte in polymeric networks. To introduce the sacrificial dynamic super-molecular cross-links, the mono-valent Na + cations can be exchanged with multivalent ones by infiltration of the latter in solution. 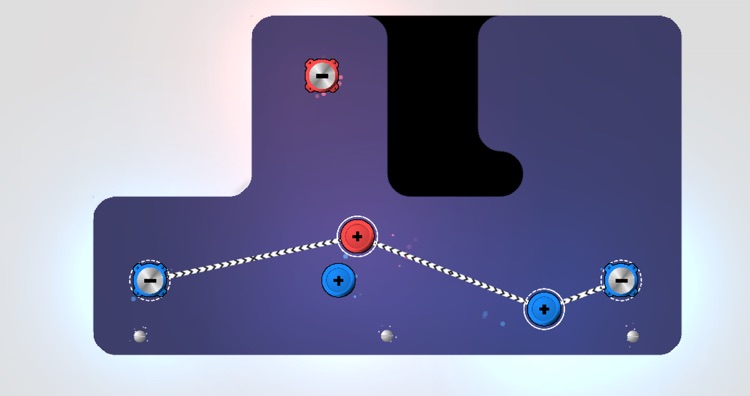
Na + cations are the major counter-ions in charged montmorillonite clay (CMC)/MTM nacre-mimetic composite due to the use of Na + CMC and Na + MTM. designed nacre-mimetic composite films from positively charged PDDA and negatively charged montmorillonite (MTM) platelets by layer-by-layer (LBL) technique. Thus tuning nanoscale ionic interactions can tailor macroscopic mechanical performance. Different from the covalent bonds and hydrogen bonds, ionic bonds can be tuned by photo-induced valence changes or via electrochemistry. Ionic bonds are versatile in terms of the valence and size of the ion, and there is a large range of dynamic bond situations. The force between the oppositely charged ions is called the electrostatic forces, therefore, ionic bonds are also called electrostatic bonds. Sacrificial ionic bonds can be constructed between oppositely charged organic and inorganic components in organic/inorganic composites. Imtiaz Hussain, Guodong Fu, in Self-Healing Polymer-Based Systems, 2020 13.12.1 Sacrificial ionic bonds In addition, the electrons are closely held in place and no charge transfer is possible, making ionic materials poor heat and electricity conductors. Bonding energy is generally high, ranging between 6 kJ/mol for example, ionic bond strength for NaCl (i.e., Na + Cl −) is about 770 kJ/mol. In general, this bond is nondirectional and has equal strength in all directions. Hence, cations and anions are bonded together via strong electrostatic attraction, forming the ionic bond. The atoms that yield electrons become positively charged ions (i.e., cations), and the ones that receive them become negatively charged ions (i.e., anions). In this case, one or more valence electrons are transferred from one atom to another one to regain the most stable configuration (i.e., eight electrons). In fact, ionic bond is formed between atoms with a high difference in electronegativity values one of the atoms has, in the outer energetic level, a few electrons (e.g., one or two electrons) and the other atom lacks of the same number of valence electrons to reach eight (i.e., a more stable configuration). 1.4) are formed when one atom donates one or more electrons to form a cation, and another atom accepts the electrons to form an anion. Since then, a wide range of ionic binding carriers have been used, and the specifics are addressed in Wisema’s Handbook of Enzyme Biotechnology. Catalase was immobilized on DEAE-cellulose in this study. Mitz was the first to announce the immobilization of an enzyme using this process in 1956. 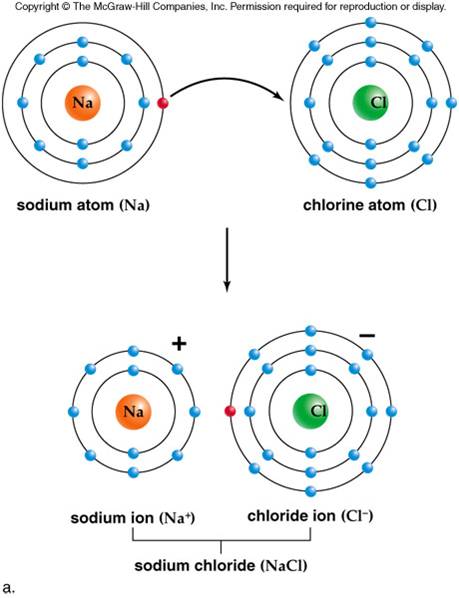 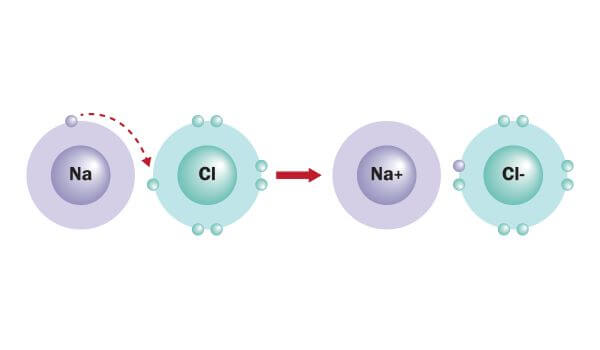 
Although it is simple and only involves simple binding reversal inputs, the drawback of this process is that it causes enzyme desorption when the pH, electrolyte concentration, or temperature changes. Ionic bonding is performed using polysaccharides and synthetic polymers with ion exchange residues ( Fig. Ionic bonding and physical adsorption vary mostly in the strength of the enzyme-support bonds, which are much stronger for ionic bonding but weaker than covalent bonding. In some cases, both ionic binding and physical adsorption can occur at the same time ( Zhu, Chen, Shao, Jia, & Zhang, 2020 ). Ionic binding immobilization is a simple reversible process that relies on the ionic binding of enzyme molecules to solid supports with ion exchange residues.
0 Comments
Read More
Leave a Reply. |
 RSS Feed
RSS Feed
